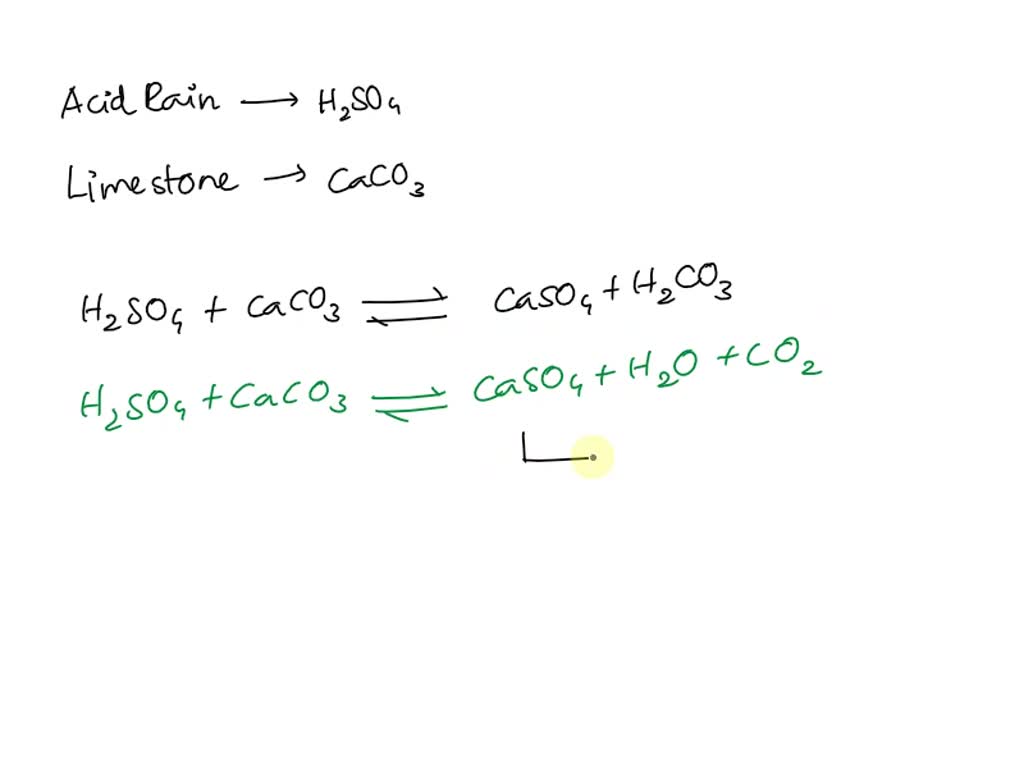
SOLVED: Acid rain is a dilute solution of acids that dissolve the calcium carbonate in limestone statues. Concentrated acids can dissolve a large piece of limestone in a few days. Statue breakdown

Toward the Mechanistic Understanding of the Additives' Role on Ammonium Nitrate Decomposition: Calcium Carbonate and Calcium Sulfate as Case Studies | ACS Omega

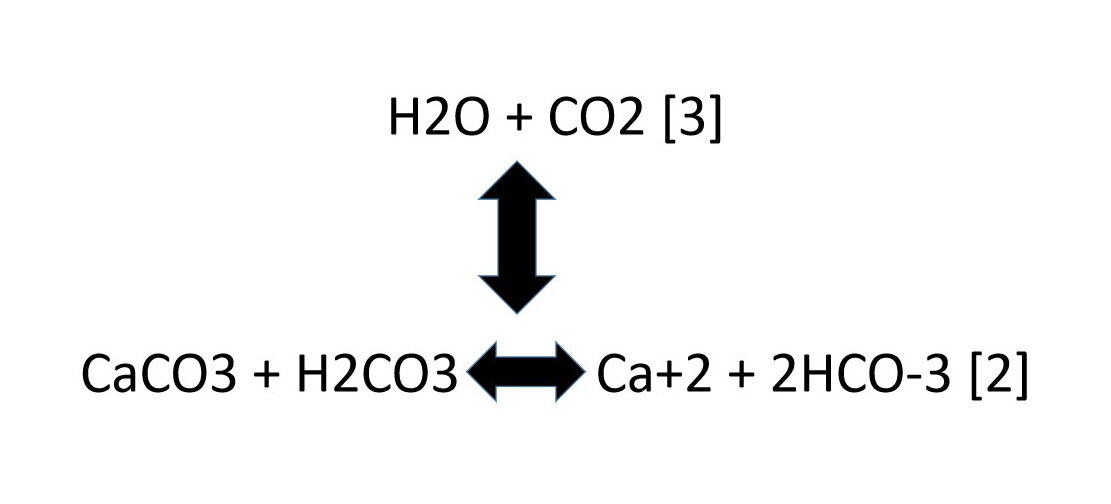
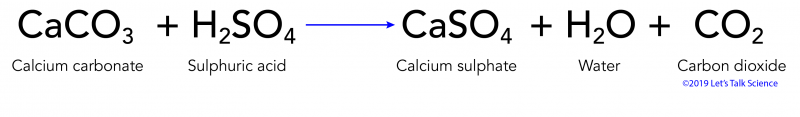
SOLVED: Write an equation to show how sulfuric acids in acid rain reacts with marble and limestone. (Both marble and limestone are primarily calcium carbonate.)

Acid Deposition Lake Barkevatn in Norway used to have healthy stocks of trout and perch. As a result of acid rain, the trout stock died out in the mid-1970s. - ppt download

Effect of simulated acid rain on the stability of calcium carbonate immobilized by microbial carbonate precipitation - ScienceDirect

CWK CWK Acid Rain State the adverse effect of these common pollutants on buildings and why these pollutants are of global concern Relate the effects. - ppt download

Everyday acid and base reactions. Calcium carbonate and rocks. Limestone is also largely composed of calcium carbonate. Bath Stone (Greater Oolite) is. - ppt download
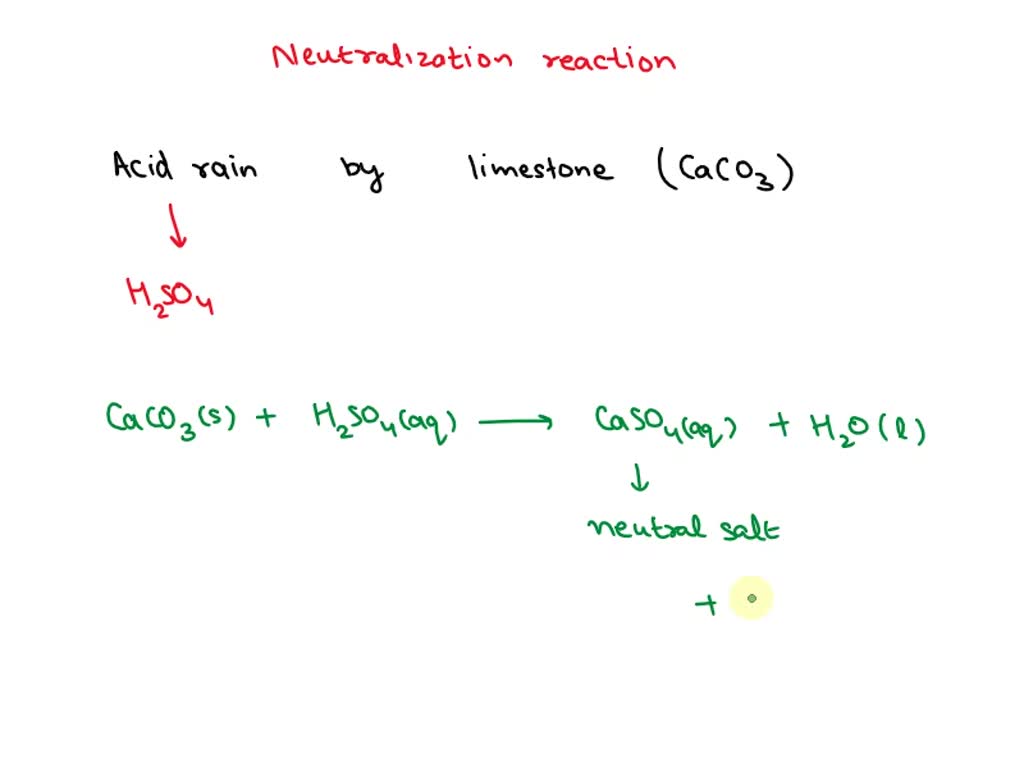
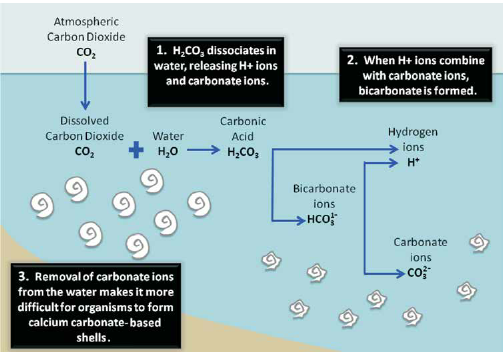
SOLVED: Acid rain is no threat to lakes in areas where the rock is limestone (calcium carbonate), which can neutralize the acid. Where the rock is granite, however, no neutralization occurs. How