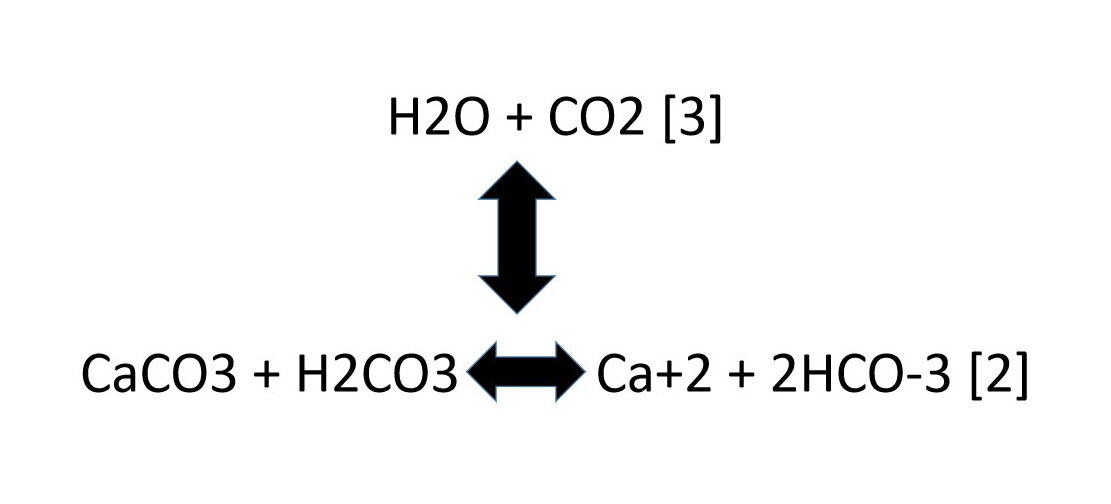
Summary of the reactions between carbon dioxide (CO2) with water (H2O)... | Download Scientific Diagram

One method of determining the proportion of calcium carbonate in a coral is to dissolve a known mass of the coral in excess acid and measure the volume of carbon dioxide formed.
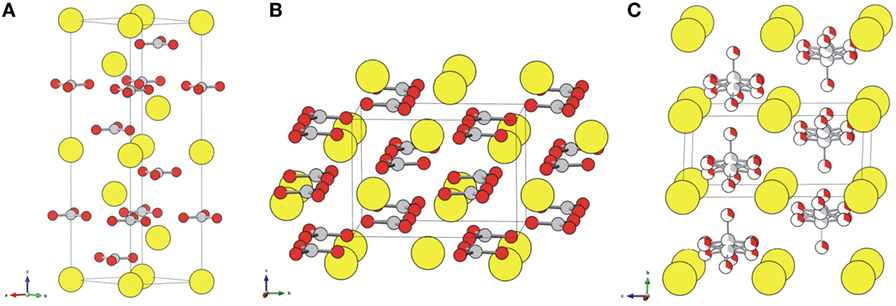
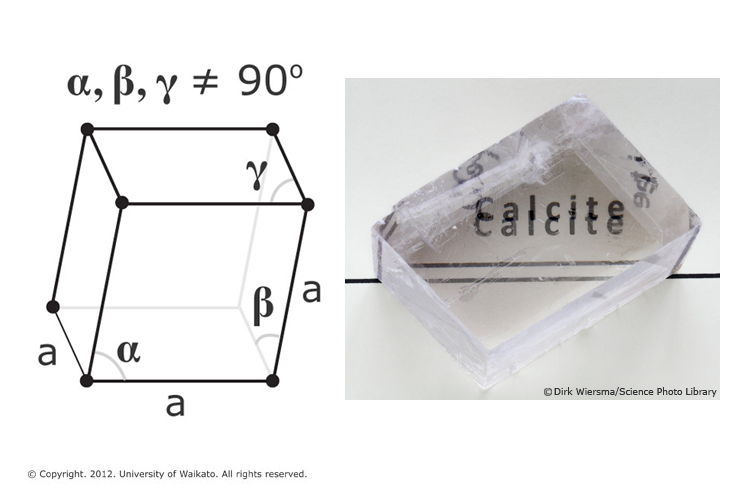
Frontiers | Calcium Carbonate Precipitation for CO2 Storage and Utilization: A Review of the Carbonate Crystallization and Polymorphism

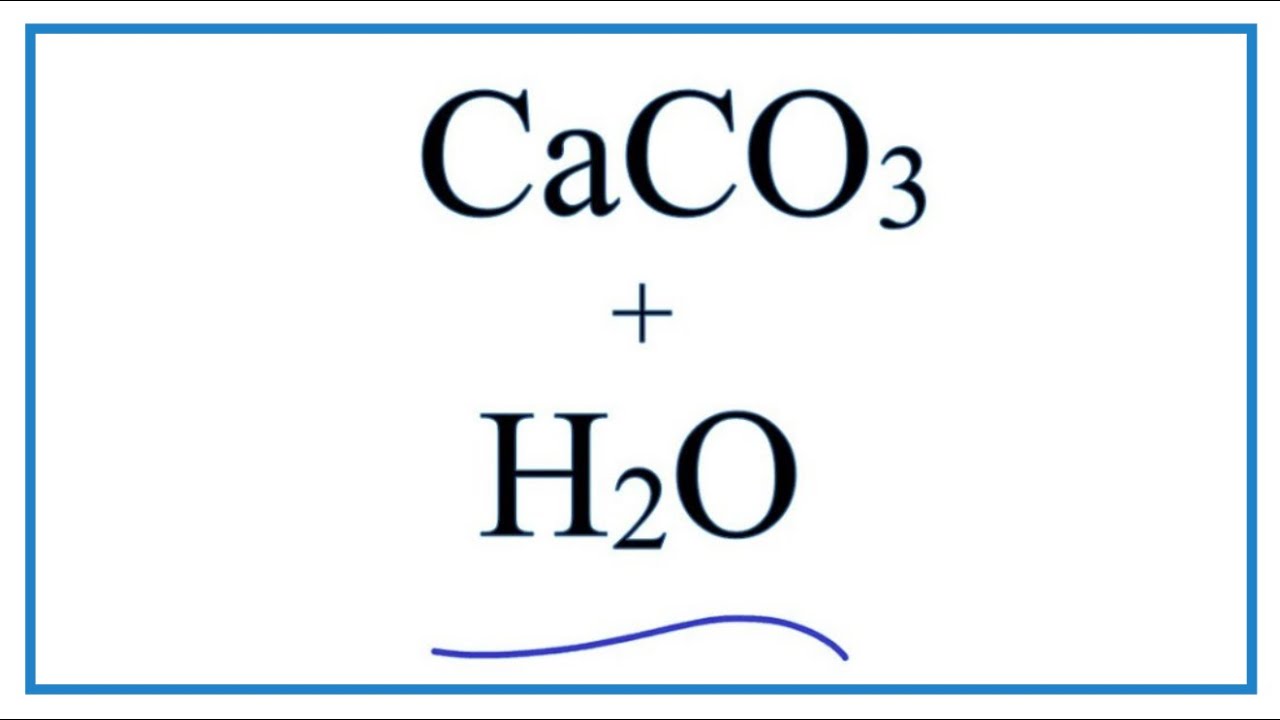
Write the balanced chemical equations for the following reactions.A Calcium hydroxide + Carbon dioxide → Calcium carbonate + waterB Zinc + Silver nitrate → Zinc nitrate + SilverC Aluminium + copper chloride
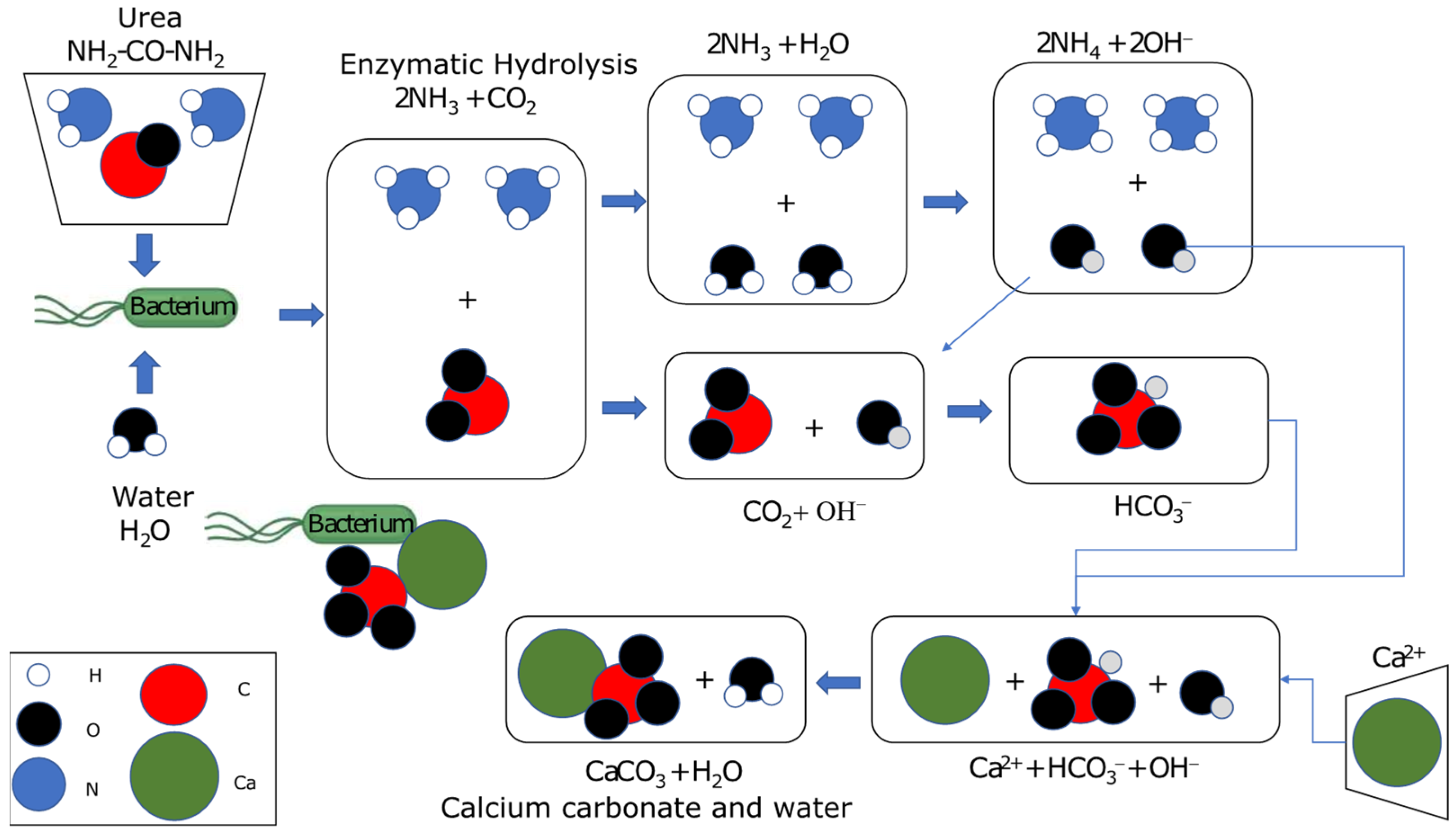
Buildings | Free Full-Text | Evaluation of the Effectiveness of a Soil Treatment Using Calcium Carbonate Precipitation from Cultivated and Lyophilized Bacteria in Soil's Compaction Water

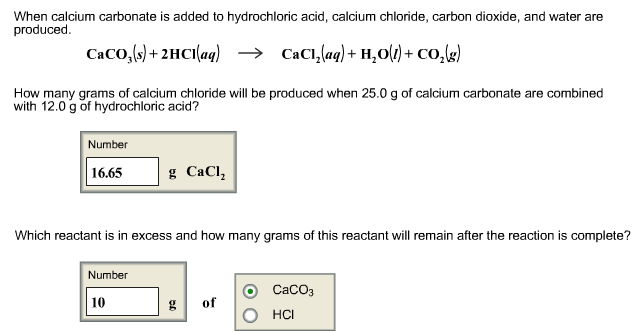
Calcium carbonate, found in limestone and marble, reacts with hydrochloric acid to form calcium chloride, carbon dioxide and water. What mass of CO2 could. - ppt download
Write the balanced chemical equations for the following reactions: i. Calcium hydroxide + Carbon dioxide → Calcium carbonate + Water - Sarthaks eConnect | Largest Online Education Community