Calcium Carbonate - Definition, Preparation Methods, Uses & Solubility - Chemistry - Aakash Byjus | AESL

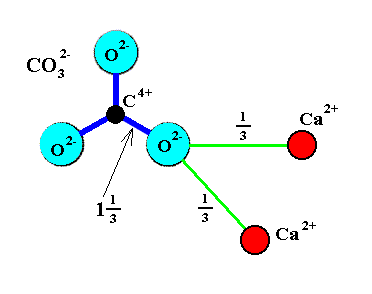
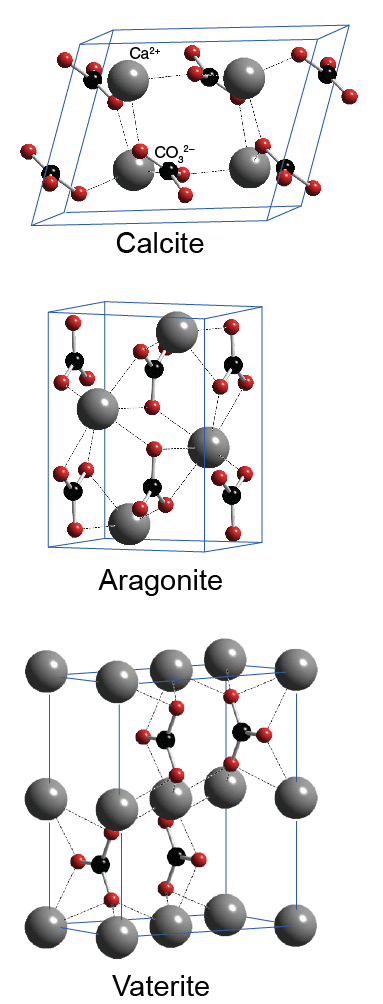
Hydrogen Bonding in Amorphous Calcium Carbonate and Molecular Reorientation Induced by Dehydration | The Journal of Physical Chemistry C

Calcium carbonate enables sustainability in polymer fiber applications | International Fiber Journal

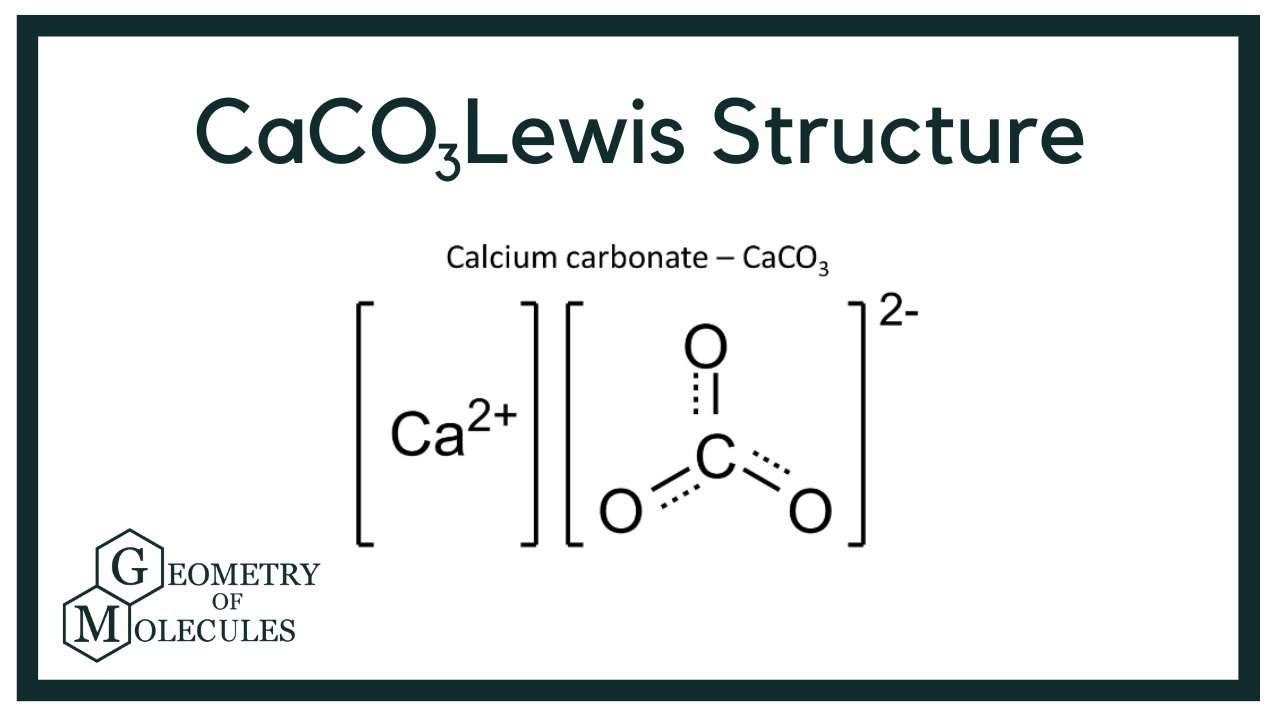
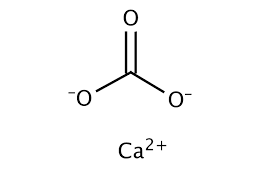
How would I go about creating a Lewis Dot structure for the Calcium Carbonate and Magnesium Hydroxide? I know how to do it for any compound with two elements such as SF5,

For calcium carbonate draw both the cation and the anions as standalone ions. Draw the most common Lewis structure, and do not draw alternative resonance forms. | Homework.Study.com
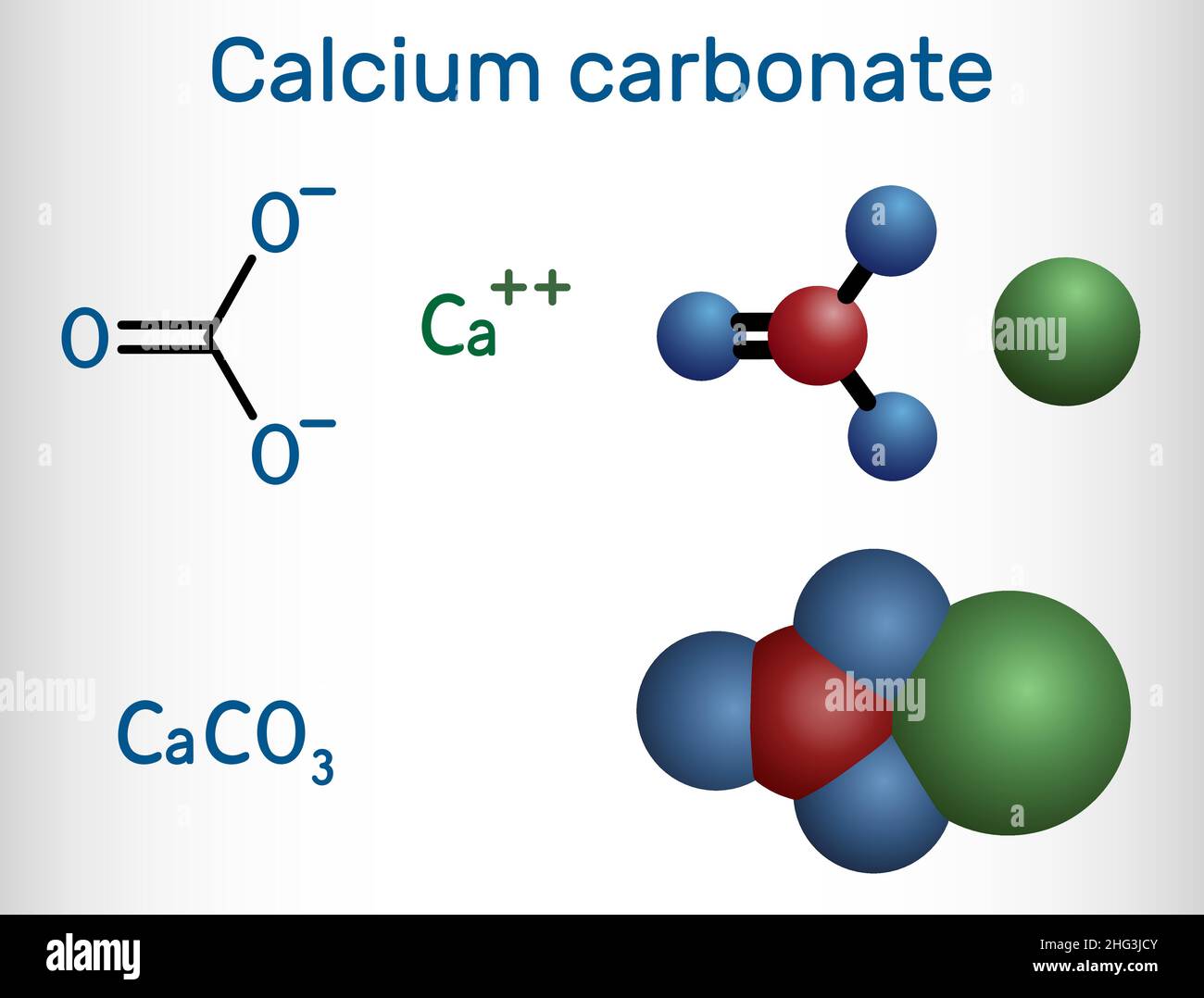
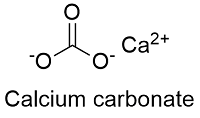
Calcium carbonate molecule. It is an ionic compound, the carbonic salt of calcium CaCO3, calcium salt, Food additive E170. Structural chemical formula Stock Vector Image & Art - Alamy