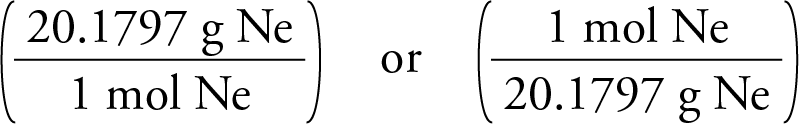a) Which molecule will have higher molecular mass?b) will they have similar/different properties?Best answer - Brainly.in

SOLVED: A mole of a substance has a mass in grams that is equal to the molecular mass. For example, a carbon atom has a mass of 12.01 u. A mole of
What's the math behind the similar numerical values of molar mass and relative molecular/atomic mass? - Quora







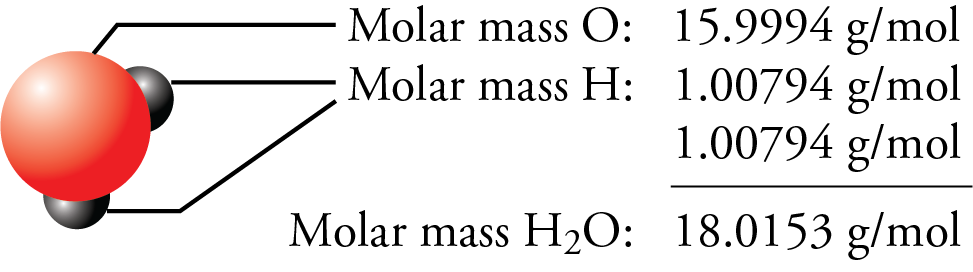



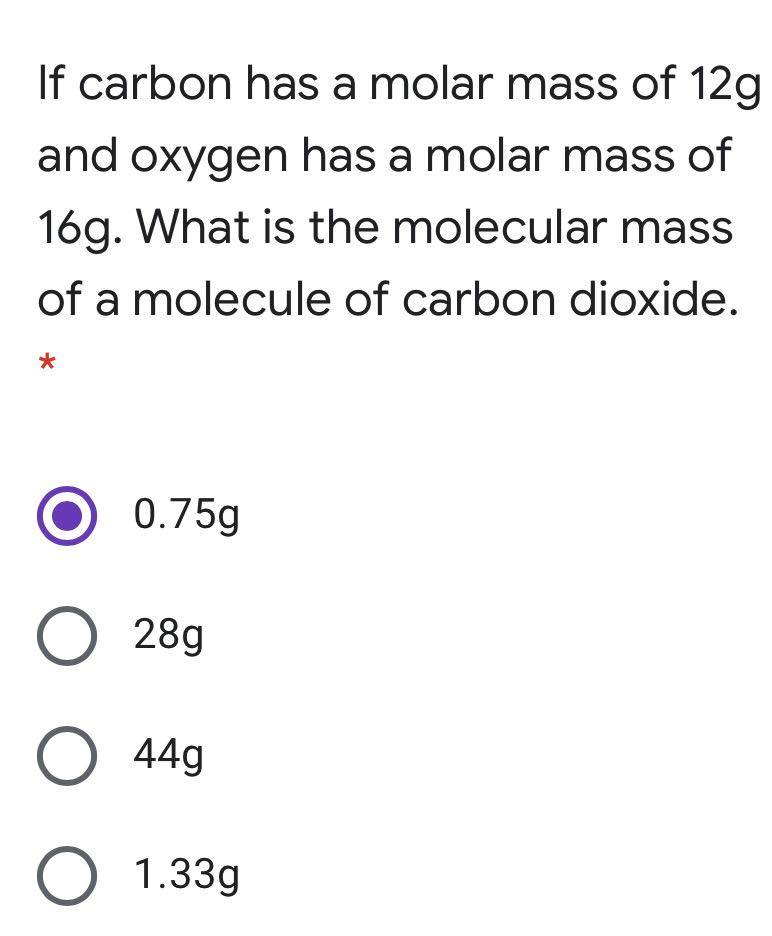



![Carbon Monoxide [CO] Molecular Weight Calculation - Laboratory Notes Carbon Monoxide [CO] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2021/11/carbon-dioxide-molecular-weight-calculation.jpg)