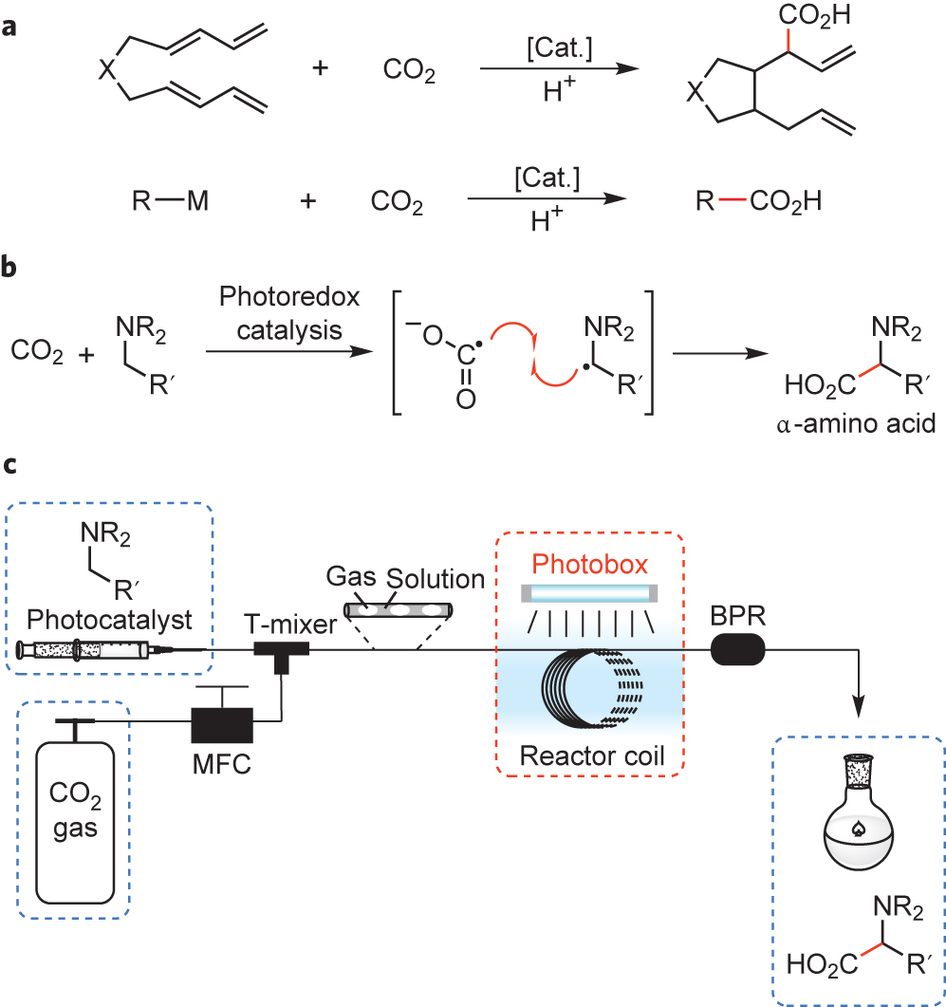
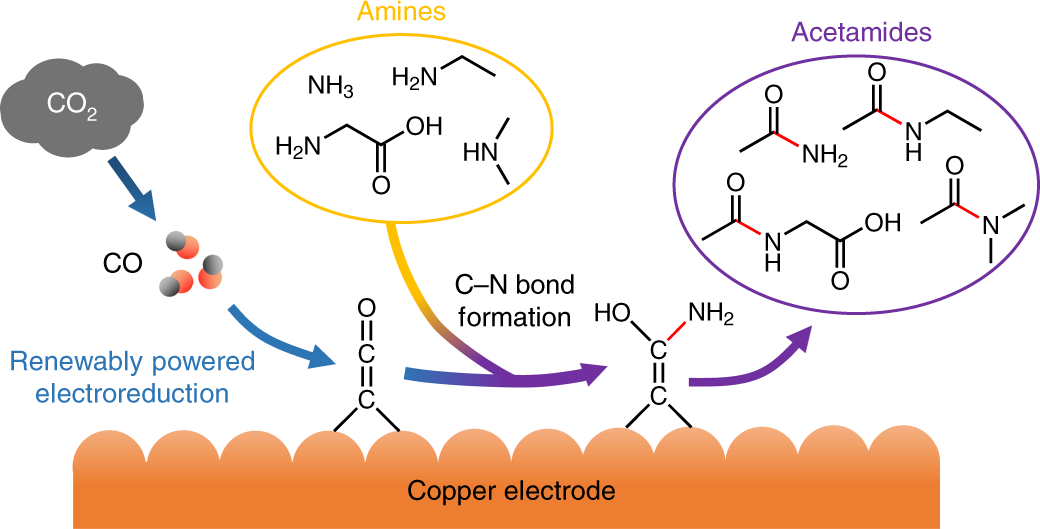
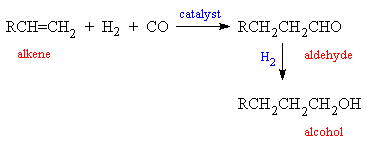The enthalpies of combustion of carbon and carbon monoxide are `-390 kJ mol^(-1)` and `-278 kJ mo - YouTube
62.The Enthalpies of combustion of carbon and carbon monoxide are 390 kJ and 278kJ respectively. The enthalpy of formation of carbon monooxide is? a) 669 kJ b) 112 kJ c) 112 kJ d) 668 kJ

Phosgene formation via carbon monoxide and dichlorine reaction over an activated carbon catalyst: Towards a reaction model - ScienceDirect
The enthalpies of combustion of carbon and carbon monoxide are -393.5 and –283kJ mol^–1 respectively. - Sarthaks eConnect | Largest Online Education Community
the enthalpies of combustion of carbon and carbon monooxide are 393.5kJ and 283kJ ,respectively the enthalpy of formation of carbon monoxide 1. 676.5kJ 2. 110.5kJ 3.110.5kJ 4.676.5

41524992Given standard enthalpy of formation of `CO(-110 \"KJ mol\"^(-1))` and `CO_(2)(-394 \"KJ mol - YouTube

The enthalpy of combustion of carbon and carbon monoxide are - 393.5 and - 283 kJ/mol respectively. The enthalpy of formation of carbon monoxide per mole is:

Reductive Cleavage of the CO Molecule by a Reactive Vicinal Frustrated PH/BH Lewis Pair | Journal of the American Chemical Society

The enthalpies of formation of CO and `CO_(2)` are `-110.5 KJ mol^(-1)` and `-393.5 KJ mol^(-1)` - YouTube
![SOLVED: The rate law for the formation of carbon dioxide from carbon monoxide and oxygen is given by: 2CO(g) + 0,(g) 2C0,(g) Rate = k [CO][02] Which one of the following mechanism SOLVED: The rate law for the formation of carbon dioxide from carbon monoxide and oxygen is given by: 2CO(g) + 0,(g) 2C0,(g) Rate = k [CO][02] Which one of the following mechanism](https://cdn.numerade.com/ask_images/736b473b932d4d048614f30fae3d5333.jpg)
SOLVED: The rate law for the formation of carbon dioxide from carbon monoxide and oxygen is given by: 2CO(g) + 0,(g) 2C0,(g) Rate = k [CO][02] Which one of the following mechanism
Formation of carbon monoxide and carbon dioxide molecules: (1) empty... | Download Scientific Diagram

The heat of formation of CO(g) and CO2(g) are Δ H = - 110 and Δ H = - 393kJmmol^-1 respectively. What is the heat of reaction (Δ H) (in kJ mol^-1 )