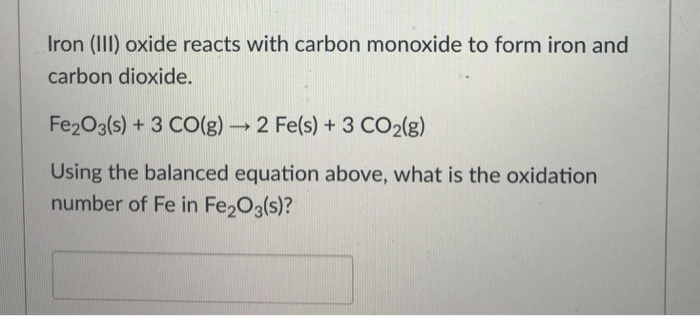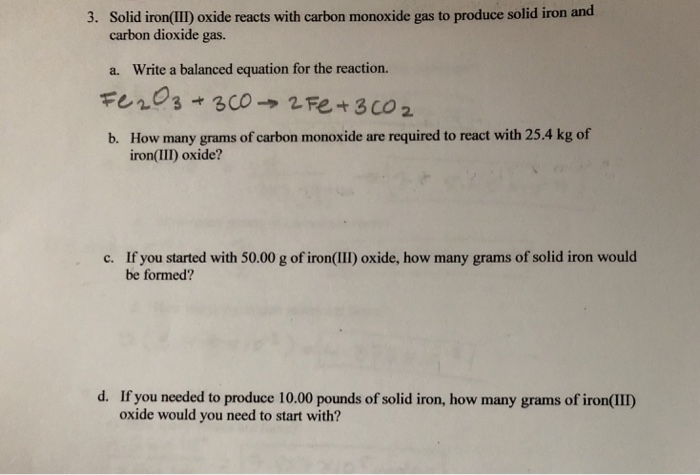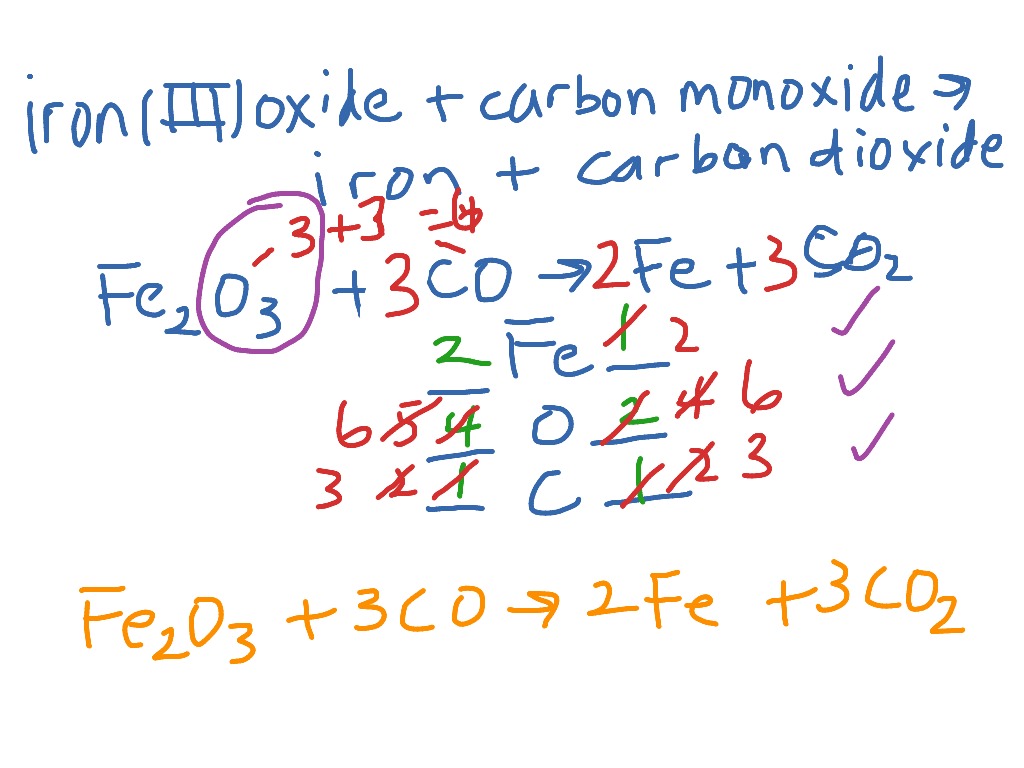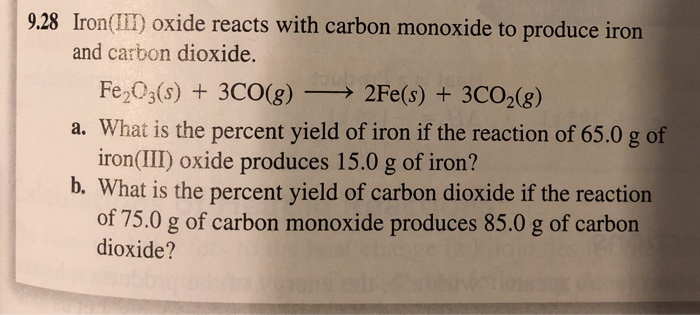Iron (III) oxide on heating with carbon monoxide gas reacts to form solid iron and liberates carbon - YouTube

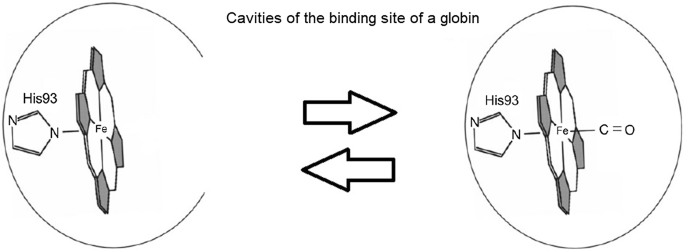
Haem catabolism by HO-1 produces equimolar amounts of carbon monoxide,... | Download Scientific Diagram

Selective reduction and homologation of carbon monoxide by organometallic iron complexes | Nature Communications

Q21 State how carbon monoxide finds application in 1 The metallurgy of iron 2 Preparation of an alco...

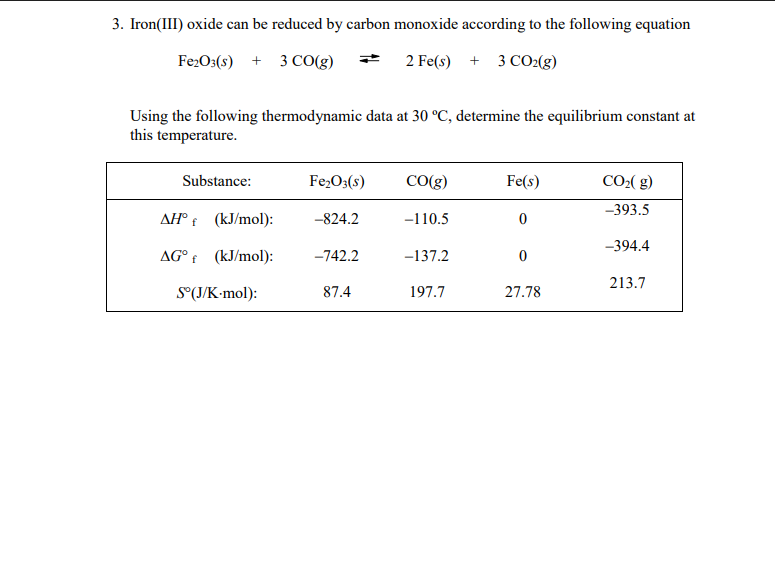
SOLVED: Pure molten Iron and carbon monoxide are produced in a blast furnace by the reaction of iron(iii) oxcide and coke (pure elementa carbon) If 2.21 kg of pure iron(iii) oxide is

Adsorption and dissociation of carbon monoxide on iron and iron-carbon clusters: Fen + 2CO and FenC + 2CO, n = 4 and 7. A theoretical study - ScienceDirect

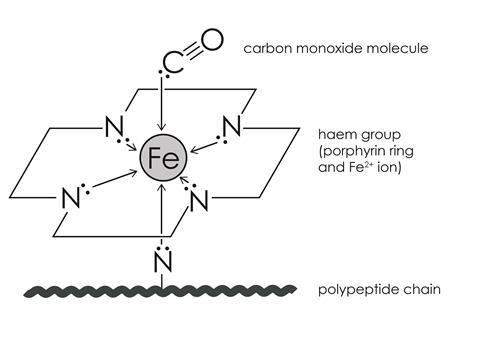
Bioactive Properties of Iron-Containing Carbon Monoxide-Releasing Molecules | Journal of Pharmacology and Experimental Therapeutics

SOLVED: Iron (III) oxide, commonly called rust; reacts with carbon monoxide to produce elemental iron and carbon dioxide Determine the enthalpy change of this reaction. (AH) (3 marks) AlIP C0-= 393.5 kJmol;

Iron(iii)oxide+Carbon monoxide=Iron+ Carbon dioxide Balanced Equation||Fe2O3+CO=Fe+CO2 Balanced Equ. - YouTube

Iron(iii)oxide+Carbon monoxide=Iron+ Carbon dioxide Balanced Equation||Fe2O3+CO=Fe+CO2 Balanced Equ. - YouTube