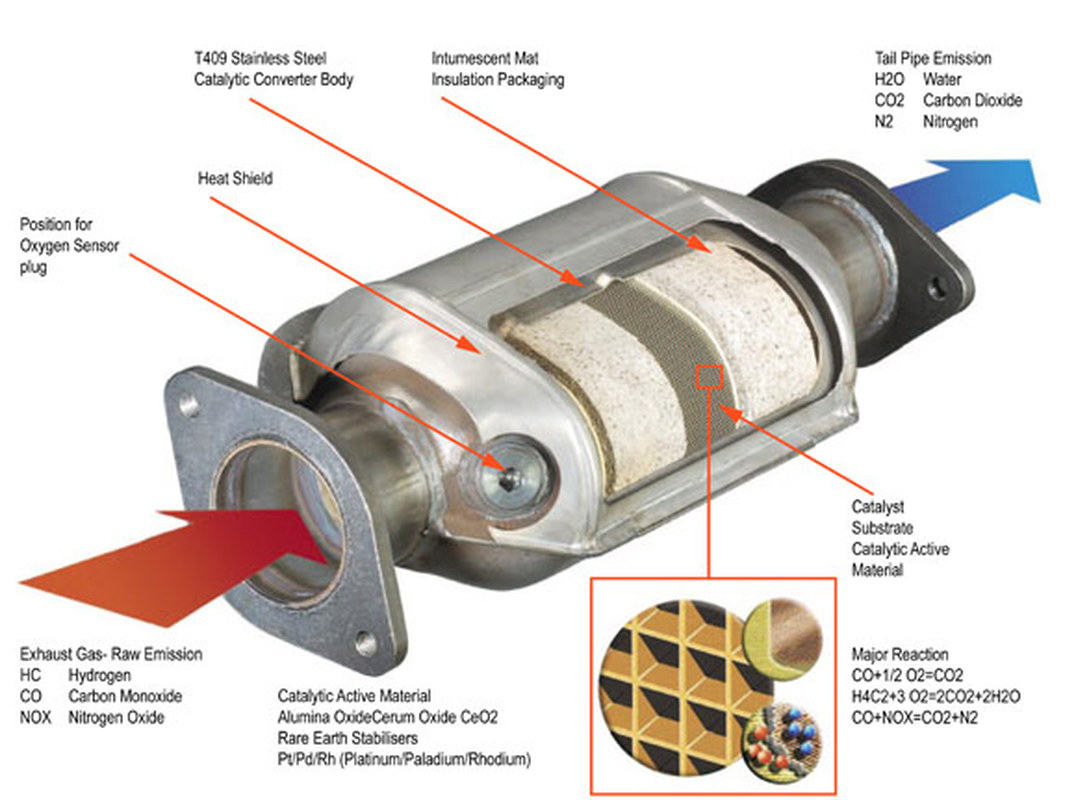
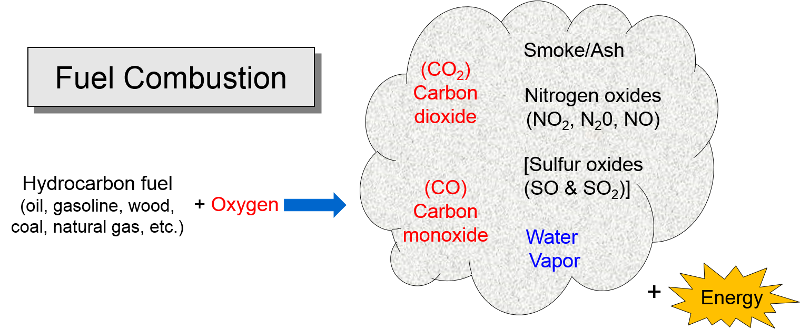
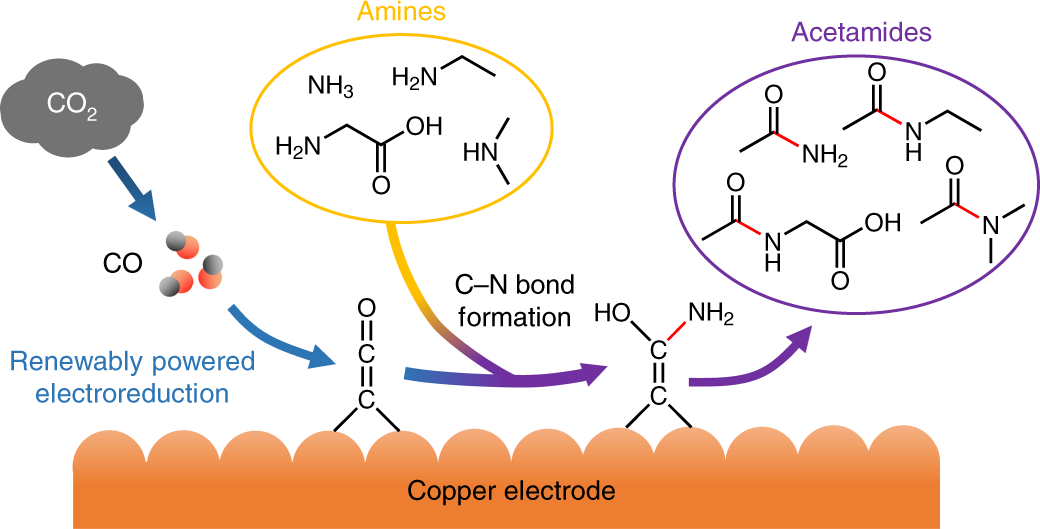
The reaction of nitrogen dioxide with carbon monoxide NO_(2)(g) +CO(g) rarr CO_(2)(g)+NO(g) has ... - YouTube

Amount of carbon monoxide (CO) and nitrogen oxides (NO x ) in smoke... | Download Scientific Diagram

SOLVED: The mechanism for the gas-phase reaction of nitrogen dioxide with carbon monoxide to form nitric oxide and carbon dioxide is thought to be NO2+NO2⟶NO3+NO NO3+CO⟶NO2+CO2 Write the rate law expected for

A review of selective catalytic reduction of nitrogen oxides with hydrogen and carbon monoxide - ScienceDirect
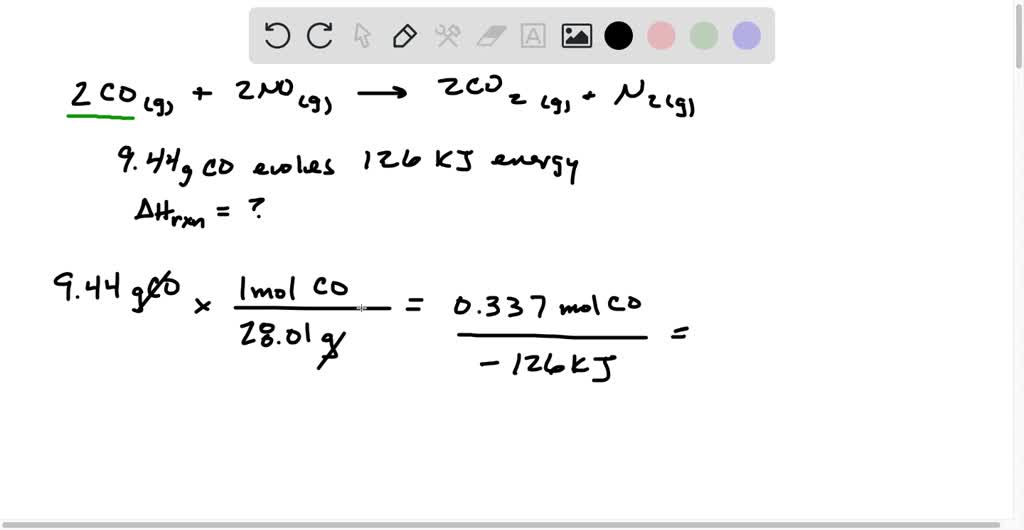
SOLVED: The reaction of carbon monoxide(g) with nitrogen monoxide(g) to form carbon dioxide(g) and nitrogen(g) proceeds as follows:2 CO(g) + 2 NO(g) 2 CO2(g) + N2(g)When 9.44 grams of CO(g) react with
✓ Solved: The mechanism for the gas-phase reaction of nitrogen dioxide with carbon monoxide to form nitric...
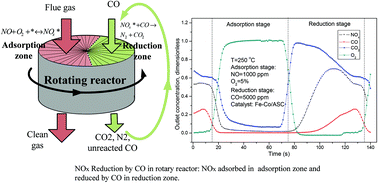
Nitrogen oxides reduction by carbon monoxide over semi-coke supported catalysts in a simulated rotary reactor: reaction performance under dry conditions - Green Chemistry (RSC Publishing)