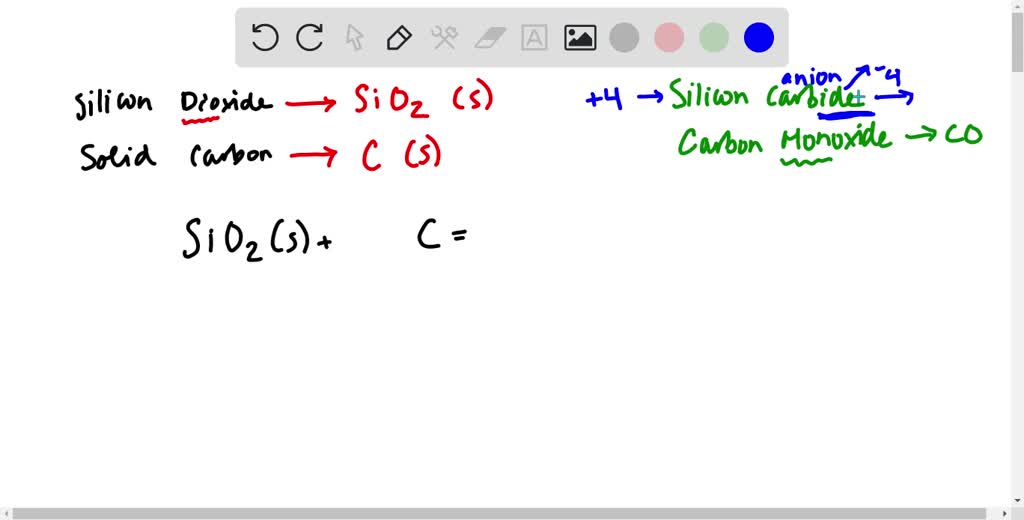
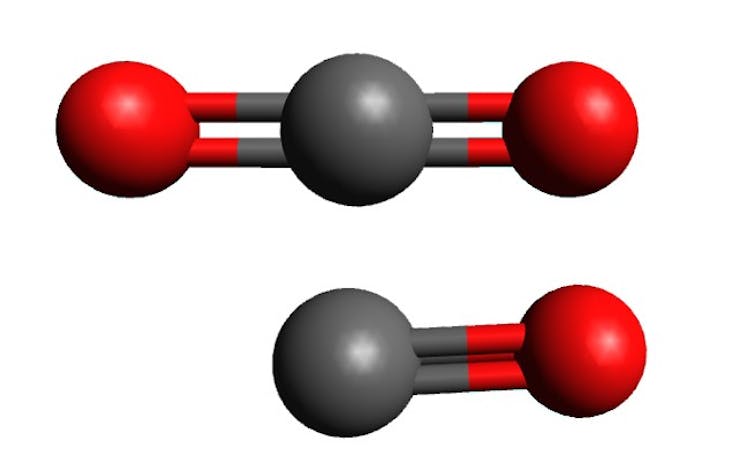
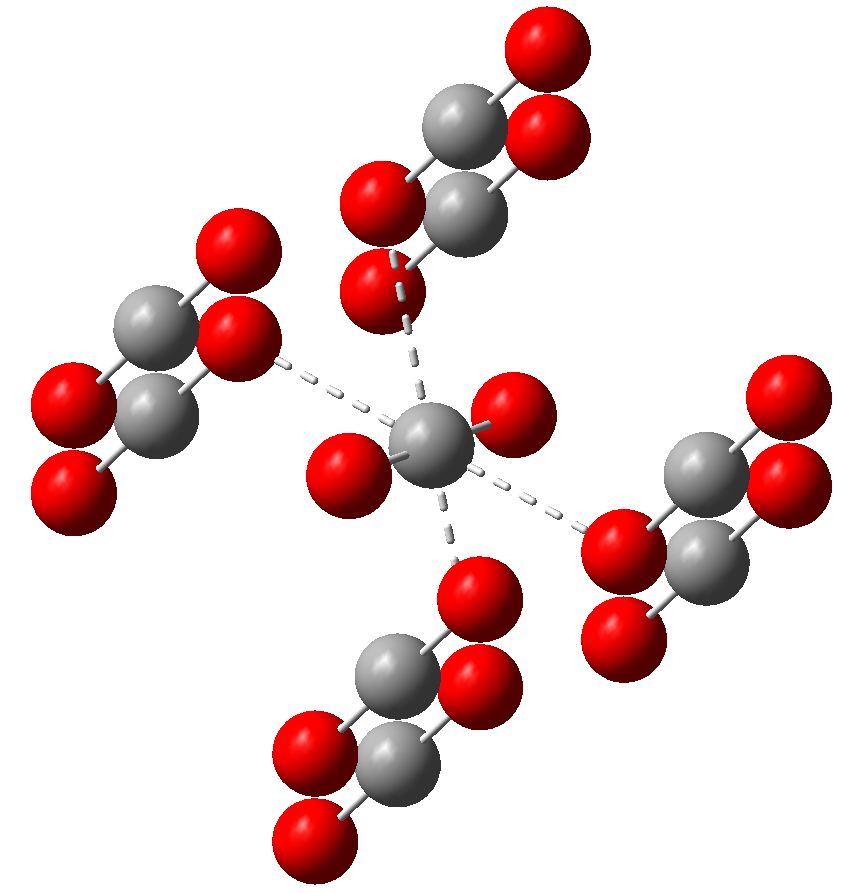
Microorganisms | Free Full-Text | Perchlorate-Coupled Carbon Monoxide (CO) Oxidation by Moorella glycerini, an Obligately Anaerobic, Thermophilic, Nickel-Dependent Carboxydotroph

Open ocean carbon monoxide apparent quantum yields (CO AQYs). Solid... | Download Scientific Diagram
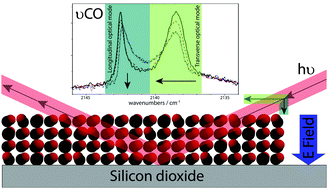
Spontaneous electric fields in solid carbon monoxide - Physical Chemistry Chemical Physics (RSC Publishing)

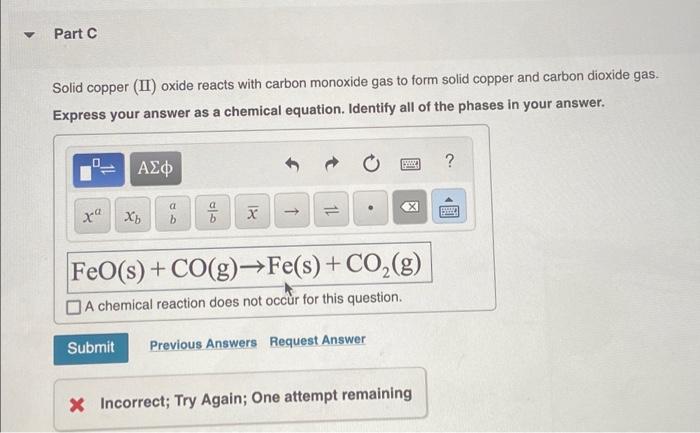
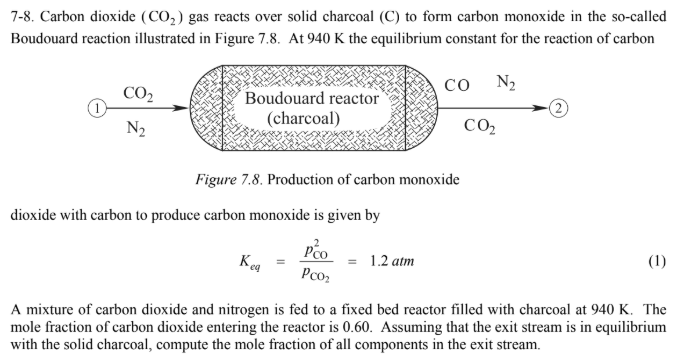
SOLVED:An equilibrium mixture contains 0.60 g solid carbon and the gases carbon dioxide and carbon monoxide at partial pressures of 2.60 atm and 2.89 atm, respectively. Calculate the value of Kp for