UV/Chlorine Process: An Efficient Advanced Oxidation Process with Multiple Radicals and Functions in Water Treatment | Accounts of Chemical Research

SOLVED:Carbon tetrachloride is produced by passing chlorine gas over carbon disulfide in the presence of a catalyst. The reaction also produces S2 Cl2 . Write the balanced reaction and identify which element

The valency of carbon is 4 and that of chlorine is 1. What will be the formua of carbon tetrachlorid - YouTube

How is covalent bond is formed? - A Plus Topper #FormationofCovalentBond | Covalent bonding, Bond, Form example

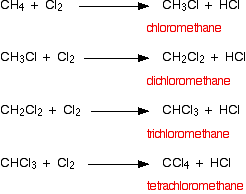
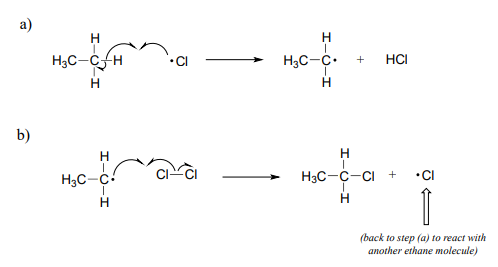
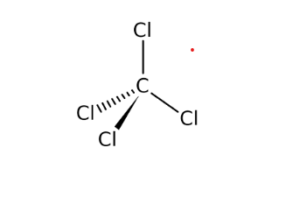
Carbon reacts with chlorine to form CCl4 . 36 g of carbon was mixed with 142 g of Cl2 . Calculate the mass of CCl4 produced and the remaining mass of reactant.