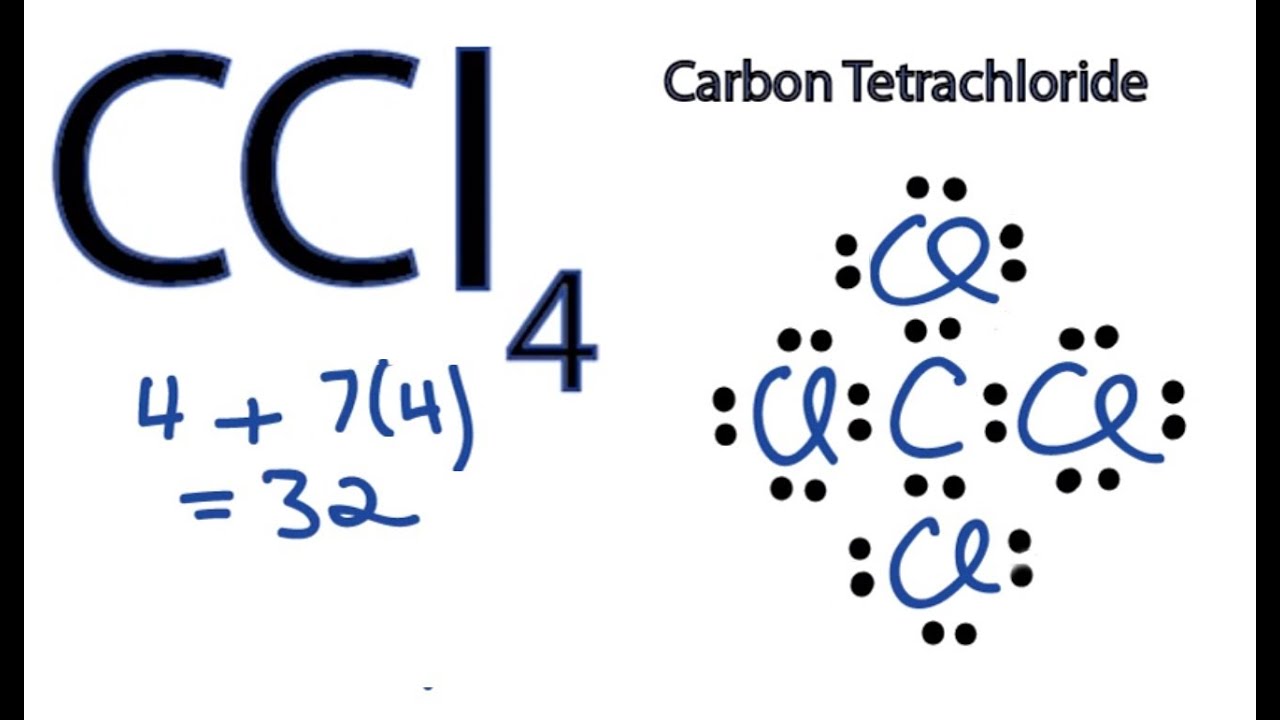
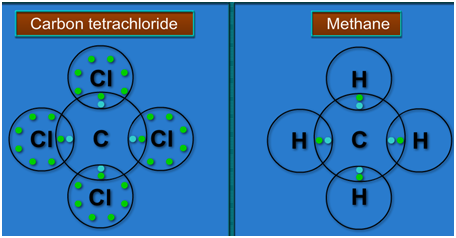
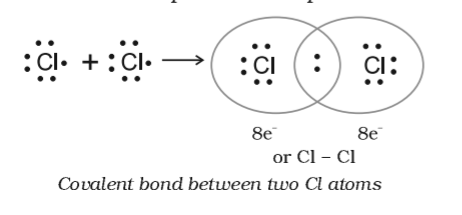
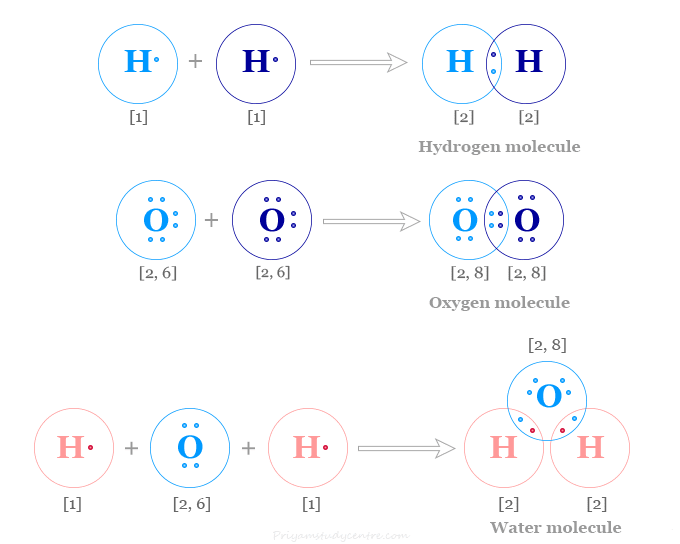
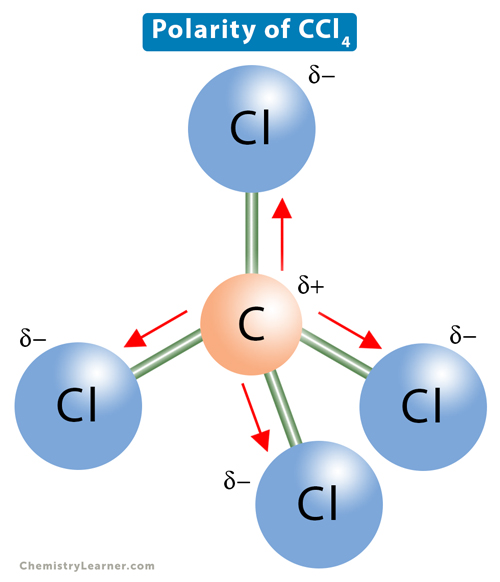
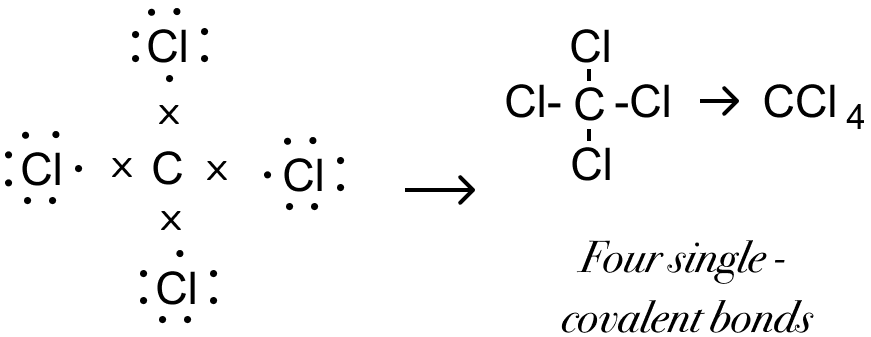
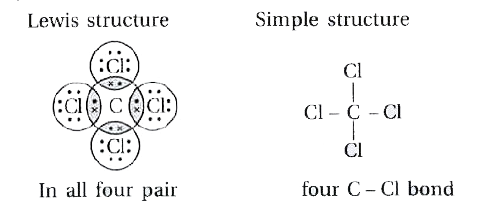Explain the formation of Covalent compound.i) Chlorine Moleculesii) Carbon Tetra Chlorideiii) Ammonia - Brainly.in
Which compound among CCL4 (carbon with chlorine) and CI4 (carbon with iodine) is more covalent and why? - Quora
Draw the structural formula of carbon tetrachloride and state the type of bond present in it - Sarthaks eConnect | Largest Online Education Community
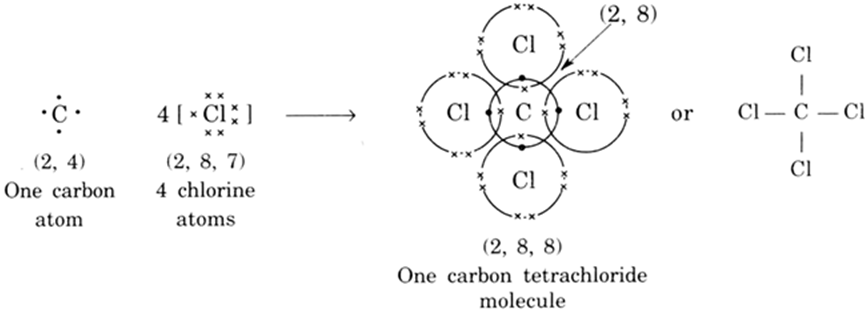
Explain the formation of covalent bonds in (i) chlorine molecule; (ii) carbon tetrachloride and (iii) ammonia. from Science Carbon and its Compounds Class 10 Jammu and Kashmir Board

Classify the following compounds as having covalent or ionic bonds: a) carbon tetrachloride b) rubidium oxide | Homework.Study.com