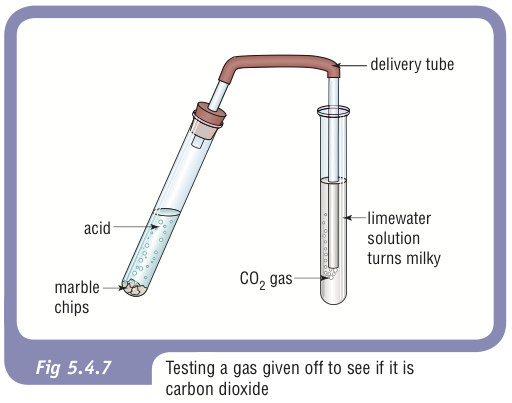
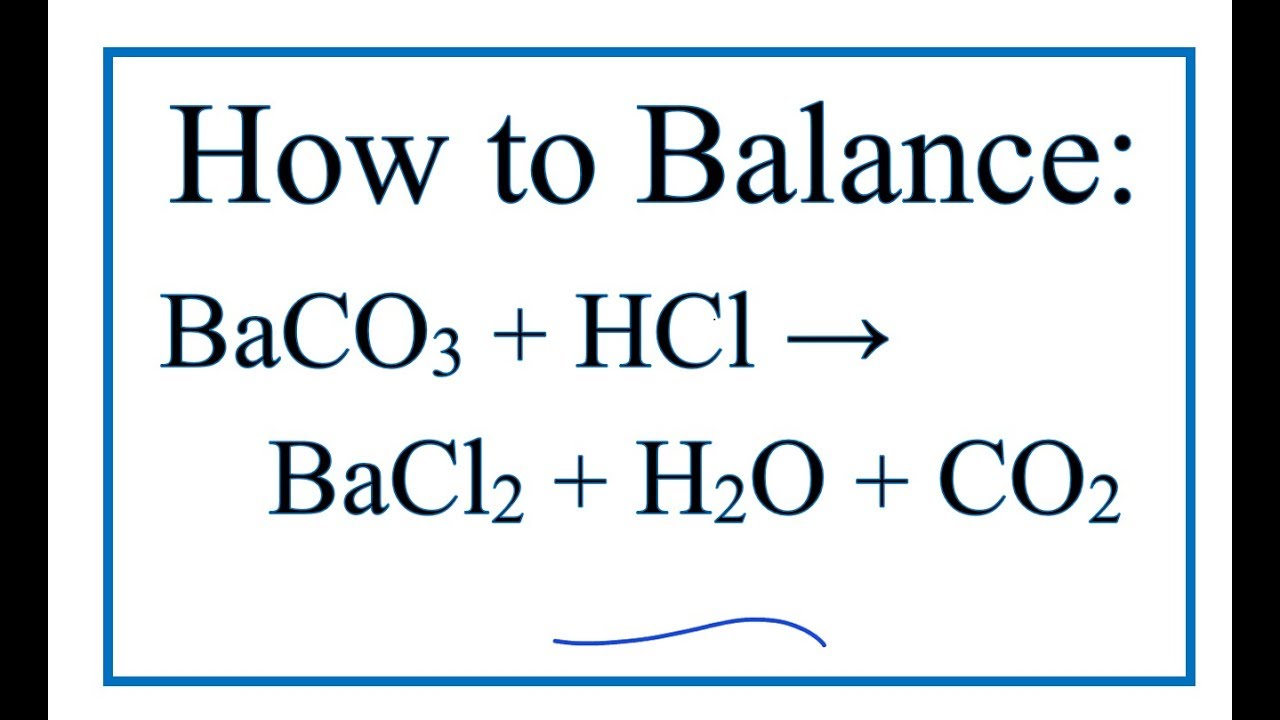
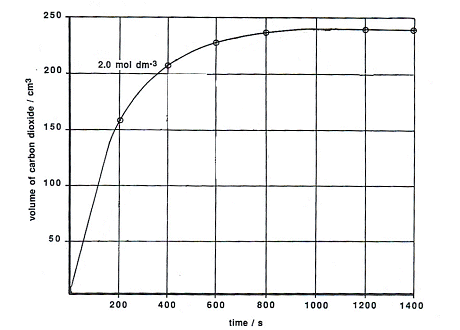
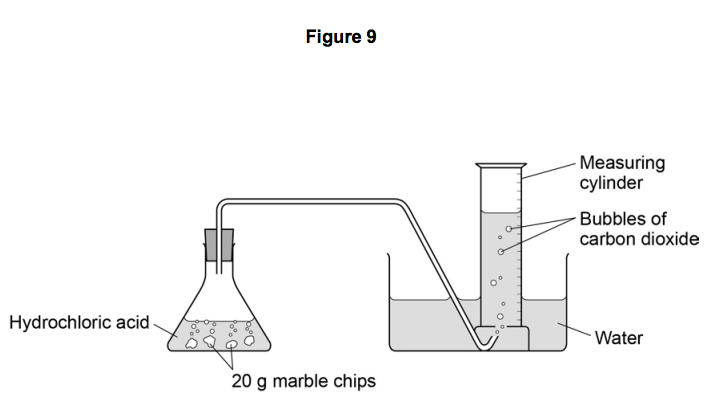
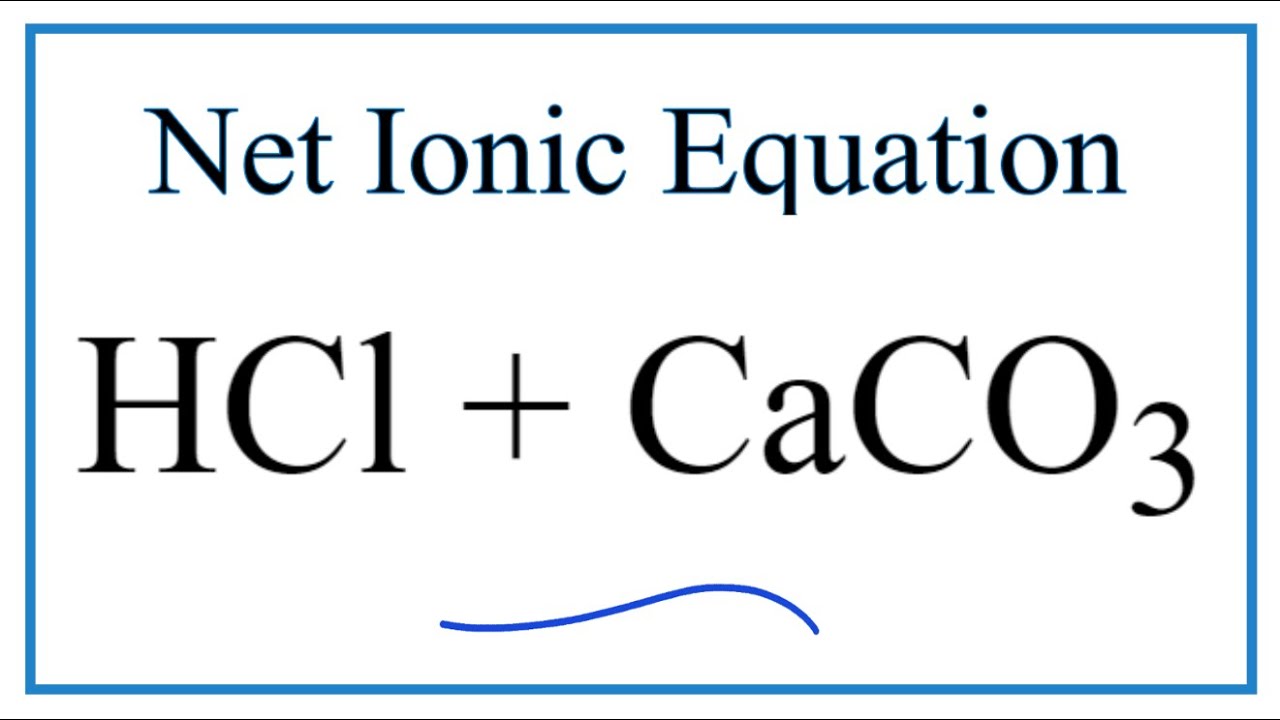35. a) Calcium carbonate reacts with aqueous HCl according to the reaction: CaCO_3(s)+2HCl(aq)→ CaCl_2(aq)+CO_2(g)+H_2O(l) . What mass of CaCO3 is required to react completely with 25ml of 0.75M HCl b) 1.0g of

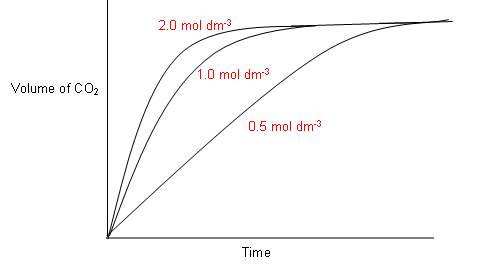
The effect of changing the concentration of hydrochloric acid on the rate of reaction with calcium carbonate - IG Exams

Question Video: Calculating the Average Rate of Reaction of Hydrochloric Acid with Calcium Carbonate | Nagwa

Net Ionic Equation for Na2CO3 + HCl | Sodium Carbonate + Hydrochloric Acid | Net Ionic Equation for Na2CO3 + HCl | Sodium Carbonate + Hydrochloric Acid Hello, Chemistry Enthusiasts! For today's
What is the pH reading after the endpoint of the neutralization reaction of calcium carbonate and HCL? - Quora

During the titration of sodium carbonate with H Cl, the dissolved carbonate ion will exist in three different forms; CO_3^{-2}, H CO_3^{-1}, and H_2 CO_3. During which part of the titration (initial,