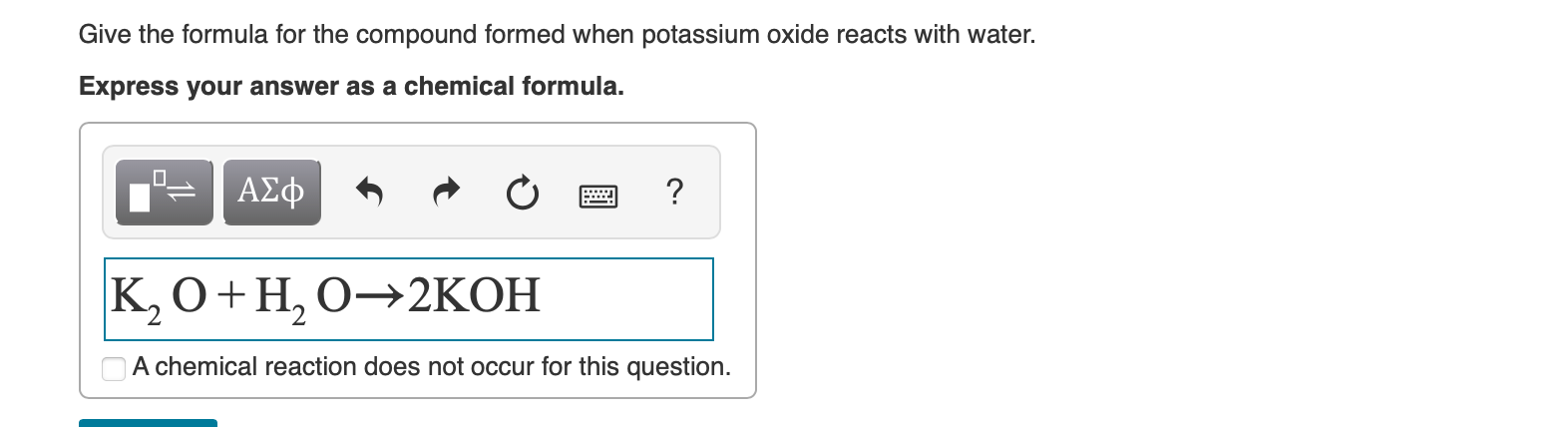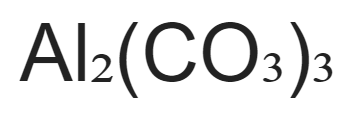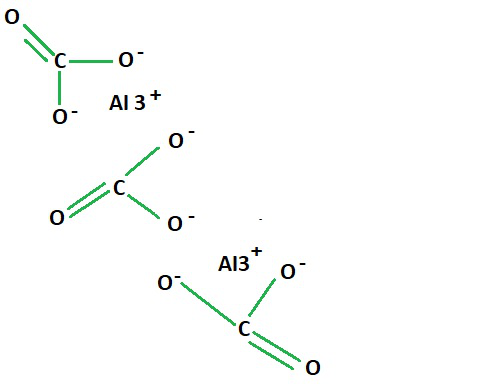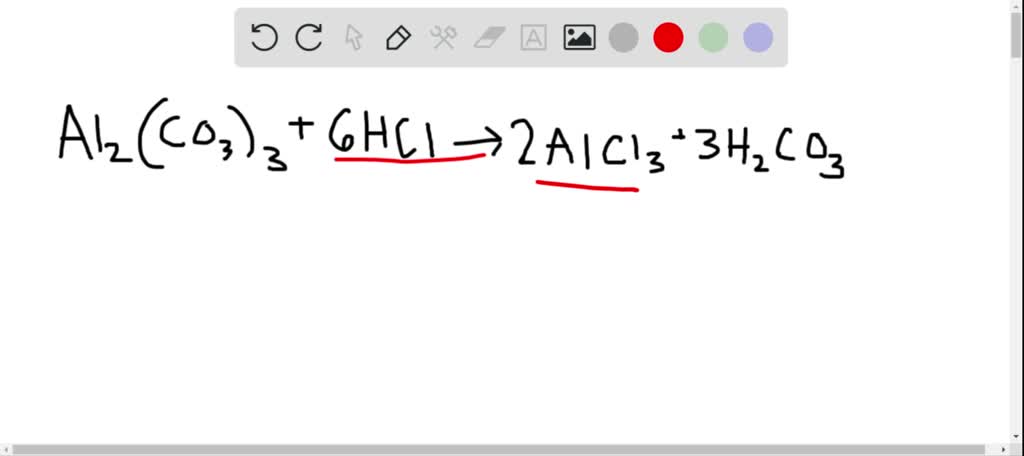
SOLVED:Aluminum carbonate is used in some antacids. Write a balanced net ionic equation for the reaction of aluminum carbonate with hydrochloric acid.

Stoichiometry Chapter 11 & 12. I. Things you should remember From the Moles Unit: Identify particles as atoms, molecules (mc), and formula units (fun) - ppt download

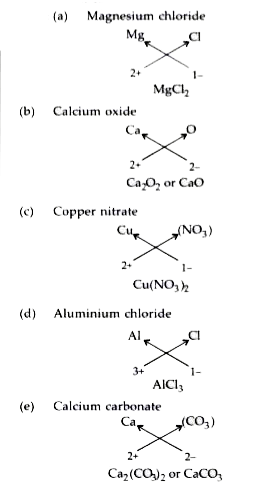
Write the formula for the following by criss- cross method :(a) Aluminium carbonate(b) Calcium hydroxide (c) - Brainly.in

Deduce the formula of: (a) Ammonium carbonate (b) Aluminium carbonate - Chemistry - Atoms and Molecules - 16513901 | Meritnation.com

How to Balance Al2(CO3)3 + HCl = AlCl3 + H2O + CO2 (Aluminum carbonate + Hydrochloric acid) - YouTube