Procedure method making insoluble salt by precipitation reaction from two soluble compounds apparatus chemicals procedures equations use of barium sulfate meal gcse chemistry KS3 KS4 Science IGCSE O level revision notes

Formation of metastable calcite-type barium carbonate during low-temperature decomposition of (Ba,Ti)-precursor complexes - ScienceDirect

Formation of Metastable Calcite-type Barium Carbonate During Low-temperature Decomposition of (Ba,Ti)- Precursor Complexes | Afribary
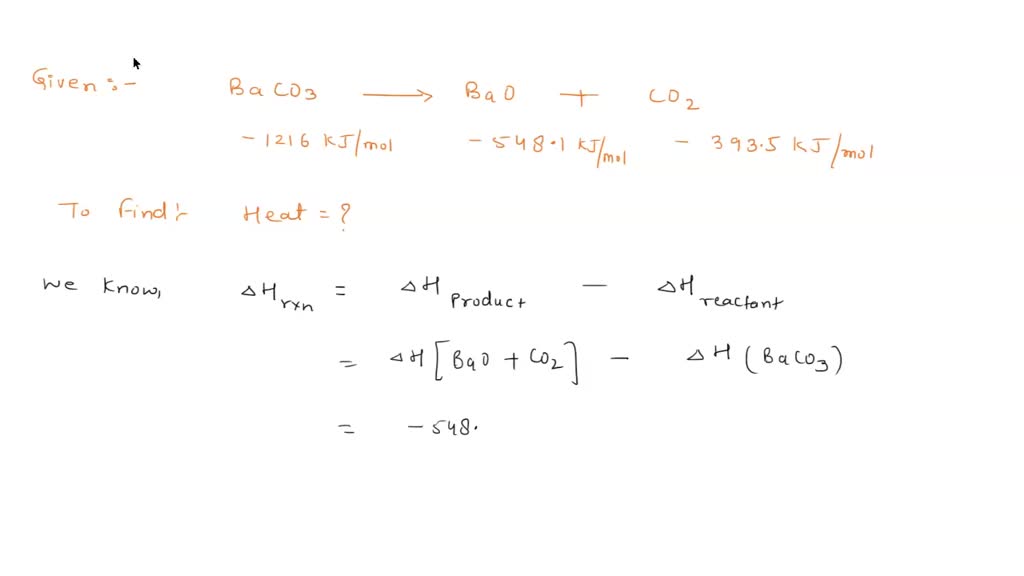
SOLVED: The Group IIA carbonates decompose when heated. For example, BaCO3(s)–>BaO(s)+CO2(g) Use enthalpies of formation and calculate the heat required to decompose 12.7 g of barium carbonate. Enthalpies of formation: BacO3=-1216.3 kj/mol

Barium carbonate decomposes when heated. BaCO3(s) → BaO(s) + CO2(g) (a) A student heated a 10.0 g sample of barium carbonate until it was fully decomposed.Calculate the volume of carbon dioxide gas




![PDF] Melting curve minimum of barium carbonate BaCO3 near 5 GPa | Semantic Scholar PDF] Melting curve minimum of barium carbonate BaCO3 near 5 GPa | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/3de18daeec30ff44d49918f1472776c04f98f970/3-Table1-1.png)